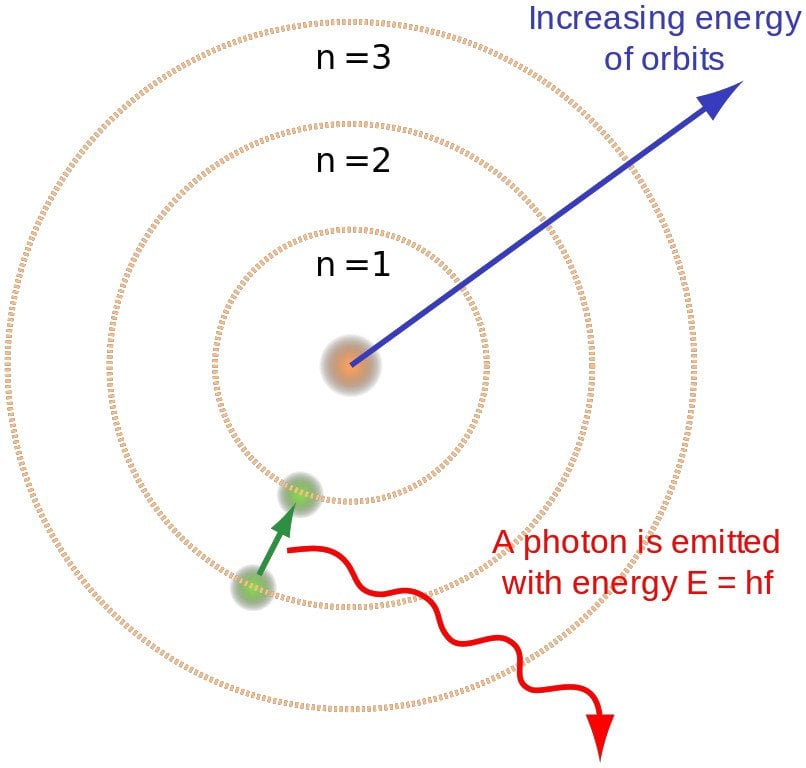 
This higher-energy, the less-stable state, is known as the electron's energized state.Īfter the excitement phase is over, the electron can regain its ground state by releasing the energy it gained. Yet, it can move to a higher-energy, less-stable level, or shell, by gaining energy. He said that the energy of an electron is quantized, which means electrons can have fixed energy levels, but, nothing in the middle.Īn electron typically involves an energy level which is called its ground state. Neils used the term energy levels (or shells) to portray these orbits of varying energy. The model shows that the electrons in atoms are in orbits of different energy levels around the core (like the planets circling the sun). 
The following are the chief postulates explained by the Neils Bohr atomic theory. Atoms gain or discharge energy when the electrons jump between the energy levels or shells. The Neil Bohr atomic model and the entirety of its replacements portray the properties of electrons with respect to a bunch of potential values. The Bohr model clarified the purpose of the Rydberg equation and justified the basic physical constants of the equation. While the Rydberg equation had been known tentatively, it didn't increase hypothetical support until the Bohr model was presented. The Bohr model's key achievement lay in clarifying the Rydberg equation for the discharge lines of nuclear hydrogen. This was an improvement to the Rutherford model and can be considered as a quantum physical explanation for it. Bohr’s Model of an Atom is a framework comprising a little, thick core encompassed by circling electrons-like the structure of the Solar System, yet with fascination given by electrostatic powers instead of gravity.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
